ChIP-Seq Analysis TutorialPart 3
|
|
In this section, we will describe the following applications:
C. Using ChIP-Seq programs with genome annotations
The ChIP-Seq server also features a collection of so-called genome annotation files, including transcription start sites collections, which are primarily used as input to the ChIP-Cor tool.
In this section we are going to illustrate four typical application examples:
- Cross-genome conservation of STAT1 binding sites
- Enrichment of STAT1 binding sites in promoter regions
- Nucleosome positioning and gene expression
- Localization Analysis of H3K4me1 domain boundaries
1. Cross-genome conservation of STAT1 binding sites
For this study, we will use the Robertson G et al., 2007 data set for in vivo STAT1 binding sites in γ-interferon stimulated HeLa cells that can be accessed as a server-resident file at the ChIP-Seq server.
We want to asses whether in vivo occupied STAT1 sites are functional or not by measuring the degree of cross-genome sequence conservation across releated species.
To allow for this type of analysis, the ChIP-Seq server offers as server-resident file, a compact SGA-formatted
version of the PhastCons genome conservation track from the UCSC genome browser database.
We will first use ChIP-Peak to generate a STAT1 peak list:
|
ChIP-Peak reports a total of 7932 STAT1 peaks.
We save the peak list both in SGA (stat1_th30_chip_peak.sga) and FPS formats (stat1_th30_chip_peak.fps) so as to be able to upload it to the FindM program of the SSA server in order to generate a new list containing the genomic coordinates of STAT1 consensus sequences occurring within 100 bp from a peak center position. The program FindM (Find Motifs) detects motifs which preferetially occur at a constrained distance from a functional site, for instance a transcription initiation site or a TF binding site.
Go to FindM and follow the instructions below:
|
You are now ready to submit the job.
We save the list of 2870 STAT1 consensus sequences extracted by FindM (findm_stat1_p.fps) and we then upload it as reference feature to the ChIP-Cor tool in order to generate the sequence conservation profile.
Go to the program ChIP-Cor and do the following:
|
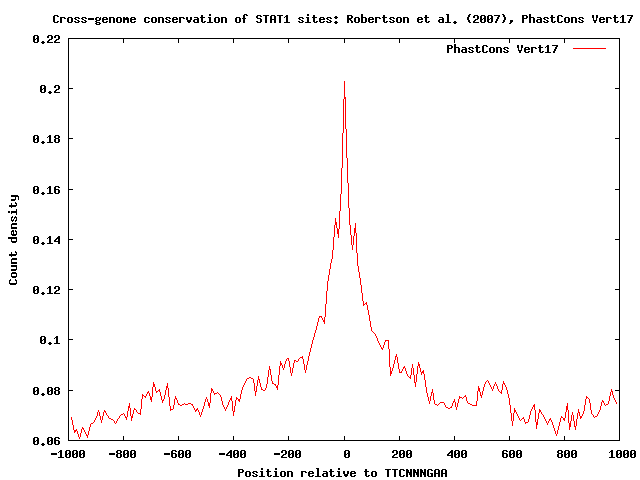
In the resulting sequence conservation profile, we observe a sharp peak centered at position zero surrounded by a larger region of increased sequence conservation, suggesting that in vivo bound STAT1 sites are indeed conserved across species, and in addition tend to occur as parts of larger conserved regulatory regions of up to 300 bp:
2. Enrichment of STAT1 binding sites in promoter regions
Another interesting topic for biologists is whether in vivo STAT1 binding sites preferentially occur in promoter regions. The ChIP-Seq server offers several transcription start site collections as server-resident files.
So, using the ChIP-Cor tool, we could easily correlate the STAT1 peak centers previously found by ChIP-Peak with transcription start site (TSS) positions to plot the STAT1 peak frequency relative to TSS.
We select the Ensembl TSS collections as reference feature, and upload the SGA-formatted output from ChIP-Peak as target feature to the ChIP-Cor tool, as follows:
|
Note that we have selected the global normalization mode, which displays the target feature abundance as fold change relative to the genome average.
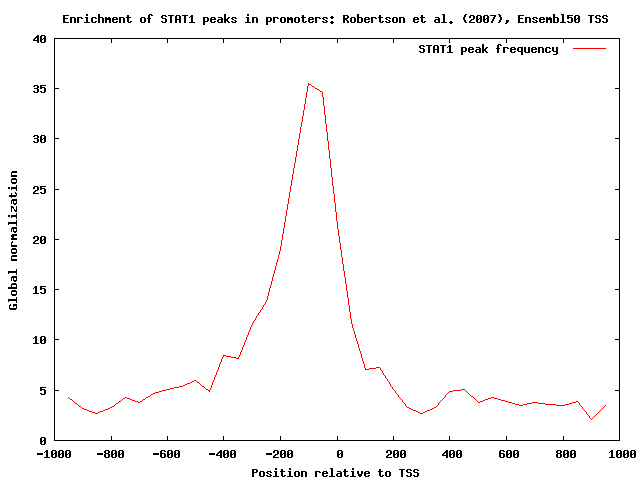
The result of this analysis shows that in vivo bound STAT1 binding sites are indeed strongly over-represented in promoter regions:
3. Nucleosome positioning and gene expression
To examine the relationship between the positioning of nucleosomes and transcriptional activity, we can use ChIP-Cor to correlate Cap Analysis Gene Expression (CAGE) tag data (T. Shiraki et al., 2003 and R. Kodzius et al., 2006) with genome-wide maps of nucleosome positions in both resting and activated human CD4+ T cells from
DE. Schones et al., 2008. Both sets of data are available on our ChIP_Seq Web Server site as server-resident files.
It is known that the positioning of nucleosomes with respect to DNA plays an important role in regulating transcription and only very recently genome-wide studies of
regulation of nucleosome phasing made it possible to correlate transcriptional
activation or repression with nucleosome reorganization.
To show an example of such results, we are going to use the ChIP-Cor tool to compare nucleosome profiles from Schones et al. with the CAGE data.
Go to ChIP-Cor and follow the instructions below:
|
You are now ready to submit the job.
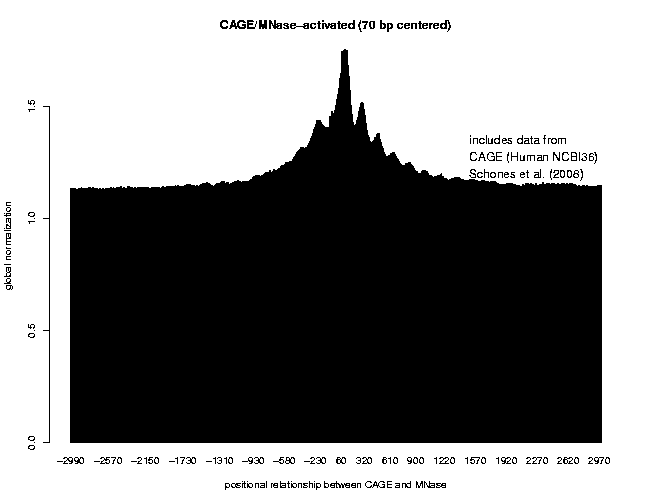
The correlation plot is the following:
Save the ChIP-Cor output file in text format (CAGE_Schones_Act_chipcor.out):
Now run ChIP-Cor for the nucleosome profile in resting CD4+ T cells from Schones et al. (hint: select feature MNase resting).
Save the ChIP-Cor output file in text format as before (CAGE_Schones_Rest_chipcor.out) and plot both output files for activated and resting T cells respectively (using gnuplot for instance):
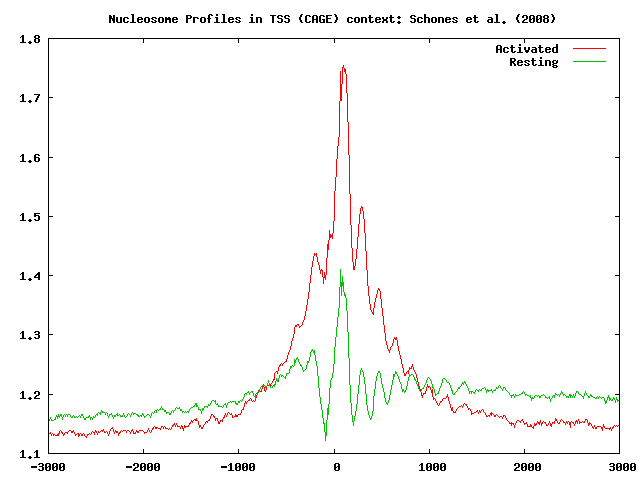
As we can see from these results, the nucleosomes near the TSS of expressed genes are phased with respect to the TSS, and that T cell activation induces nucleosome reorganization. Our data indicate that the nucleosome level does not show a big change in the region upstream of the TSS, whereas we observe a significant increase in the +1 and +2 nucleosome levels downstream of the TSS. This observation is in agreement with what has been found by Schones et al., 2008.
We previously analyzed (tutorial part 2) the genome-wide distribution patterns of histone methylation
in human CD4+ cells using ChIP-Seq (Barski et al., 2007). We observed that the H3K4me3 profiles displayed several subpeaks, each spanning approximately 150 bp, suggesting that these short sequence reads may elucidate nucleosome positions:
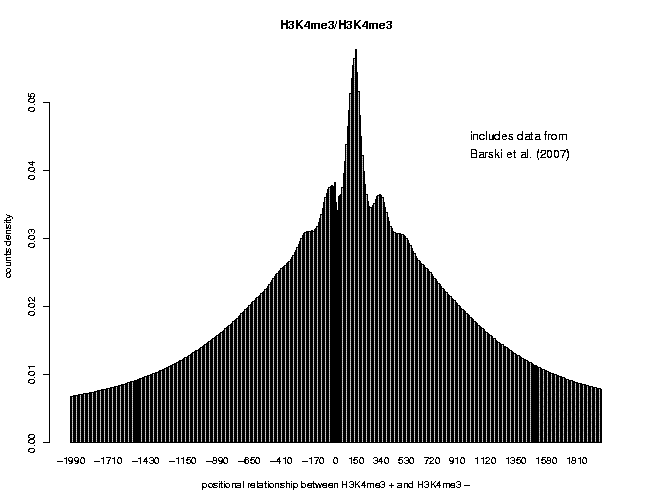
Try now to study H2A.Z deposition and H3K4me3 modification in promoter regions, as we have shown in the example above, to show how they may facilitate nucleosome eviction or repositioning in promoter regions of the human genome.
The figure below shows different nucleosome positioning patterns around TSSs:
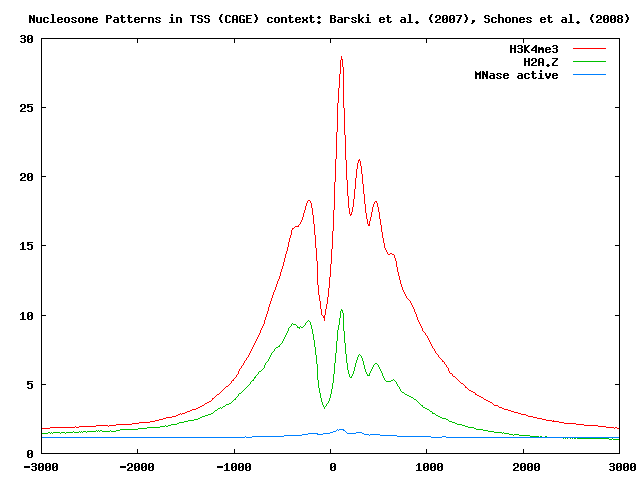
The results of this analysis show that nucleosome positioning is indeed correlated with gene expression activity.
4. Localization Analysis of H3K4me1 domain boundaries
In this section, we will try to show how the ChIP partitioning tool ChIP-Part, used in conjuction with the correlation tool ChIP-Cor, can be useful for analysing in more detail genome-wide histone modification patterns and co-localizing them with other significant epigenetic properties.
To this purpose, we choose H3K4me1, which is known to be active in transcription initiation, and we start be extracting H3K4me1 boundaries across the human genome by means of ChIP-Part.
Go to the program ChIP-Part and procede as follows:
|
The analysis revealed 35241 H3K4m1-modified regions. Save both the partitioning statistics and the SGA-formatted file:
ChIP-Part-H3K4me1.sga
ChIP-Part-H3K4me1.stats.txt
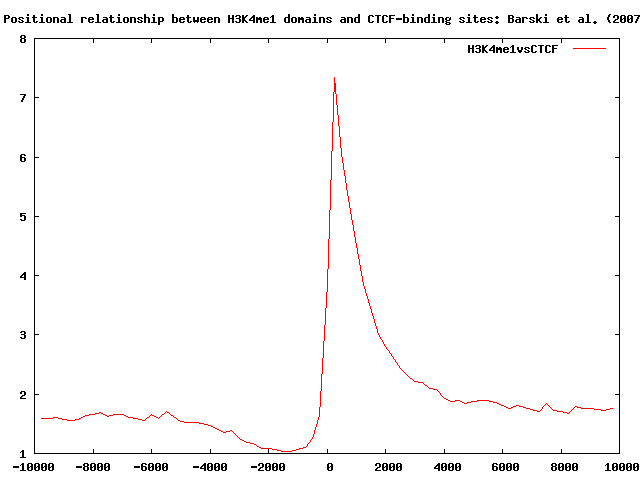
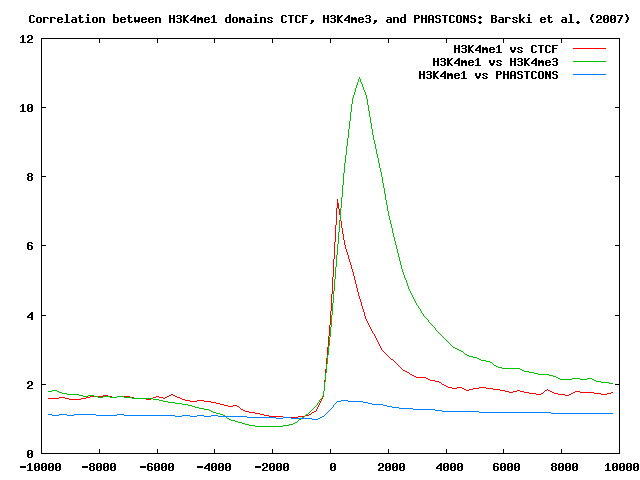
Using ChIP-Cor we now search for H4K3me1 domain boundaries (edges of H3K4me1 domains) that occur near CTCF-binding sites, and H3K4me3 marks. For completeness, we also look at cross-genome conservation maps around H3K4me1 domain boundaries.
Go to ChIP-Cor and follow the instrauctions below:
|
The histogram shows a sharp peak at about 250 bps:
Similarly, we can correlate H3K4me1 domain boundaries with H3K4me3 tag maps and cross-genome conservation scores.
Here below is a summary of the results:
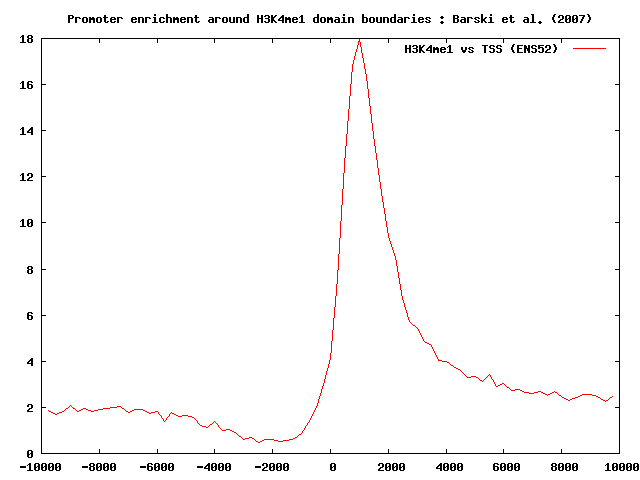
We now split the H3K4me1 chromatin domains into two different sets: promoter-enriched and promoter-depleted H3K4me3 boundaries. To do this, we should proceed as follows.
We first run ChIP-Cor to correlate H3K4me1 domain boundaries with a list of annotated promoters from the ENSEMBL52 set. We then use the Feature Extraction tool to select H3k4me1 boundaries with at least 1 transcription initiation site between -1000 bp and 2000 bp. We activate the depleted feature extraction checkbox to extract the promoter-depleted set. The promoter-enriched set contains 8925 domain boundaries whereas the promoter-depleted set contains 61557 sites:
ChIP-Cor-H3K4me1_TSS_ENS52_enriched.sga
ChIP-Cor-H3K4me1_TSS_ENS52_depleted.sga
It's interesting to look at the localization of CTCF-binding sites, H3K4me3 and PHASTCONS conservation scores in both the previously selected sets.
Perform the correlation analysis as indicated above.
Here below you can see the summary results.
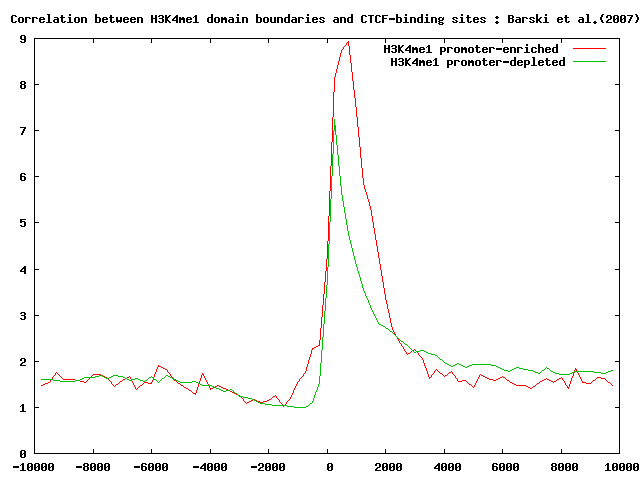
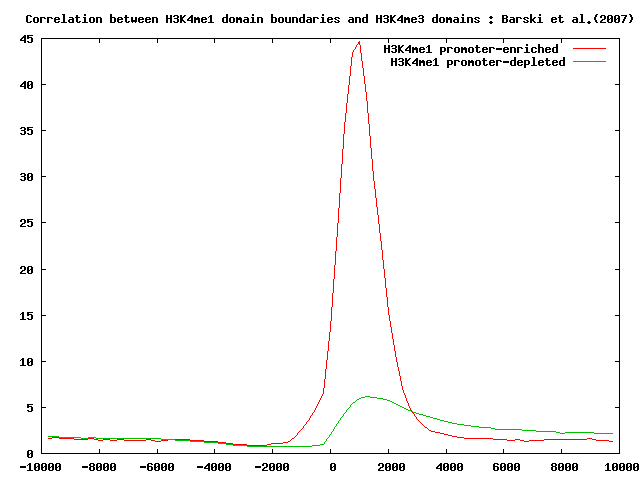
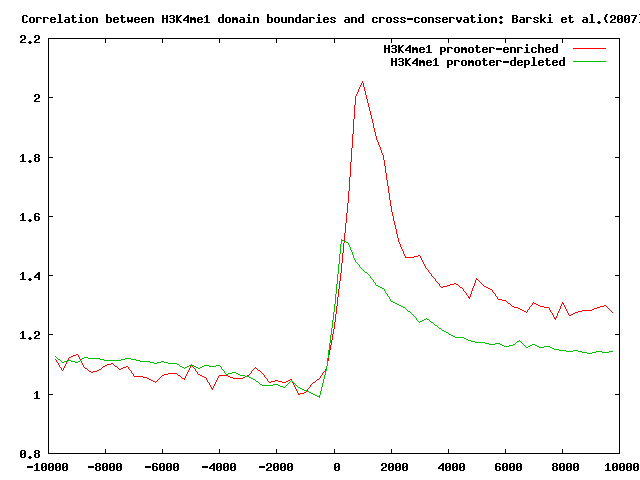
Localization analysis of H3K4me3 chromatin domain boundaries indicates that those domains that occur nearby promoter regions are conserved across different species and are strongly correlated with H3K4me3 domains.
